|
Here we further explore the nature of this state function and define it mathematically. 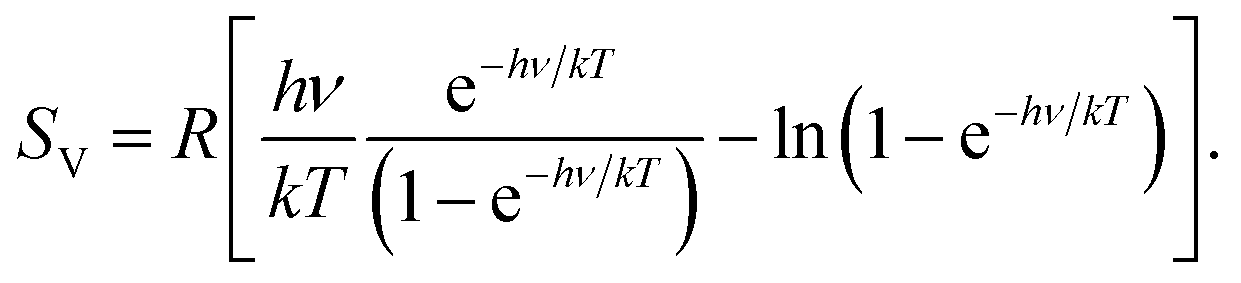
In Chapter 13, we introduced the concept of entropy in relation to solution formation. To help explain why these phenomena proceed spontaneously in only one direction requires an additional state function called entropy (S), a thermodynamic property of all substances that is proportional to their degree of "disorder". Moreover, the molecules of a gas remain evenly distributed throughout the entire volume of a glass bulb and never spontaneously assemble in only one portion of the available volume. For example, after a cube of sugar has dissolved in a glass of water so that the sucrose molecules are uniformly dispersed in a dilute solution, they never spontaneously come back together in solution to form a sugar cube. Since there is no disorder in this state, the entropy can be defined as zero. Imagine cooling the substance to absolute zero and forming a perfect crystal (no holes, all the atoms in their exact place in the crystal lattice). For a full video: see Thus enthalpy is not the only factor that determines whether a process is spontaneous. The absolute entropy of any substance can be calculated using equation (1) in the following way. When water is placed on a block of wood under the flask, the highly endothermic reaction that takes place in the flask freezes water that has been placed under the beaker, so the flask becomes frozen to the wood. entropy and to derive absolute entropy values under specific. Standard absolute entropy values are given in units of joules per kelvin per mole J K-1 mol-1 Values of standard absolute entropy (S) for many substances have been tabulated. According to the Boltzmann equation, the entropy of this system is zero. The reaction of barium hydroxide with ammonium thiocyanate is spontaneous but highly endothermic, so water, one product of the reaction, quickly freezes into slush. The standard absolute entropy of a substance, S, is the absolute entropy of a substance in its standard state (298.15 K, 100 kPa). In physics, black hole thermodynamics is the area of study that seeks to reconcile the laws of thermodynamics with the existence of black hole event horizons.As the study of the statistical mechanics of black-body radiation led to the development of the theory of quantum mechanics, the effort to understand the statistical mechanics of black holes has had a deep impact upon the understanding of. 
Our procedures are simple, robust, and reliable and can be used by specialists and nonspecialists alike.\): An Endothermic Reaction. Coupled with our published procedures, which relate volume to other thermodynamic properties via lattice energy, the correlation reported here complements our development of a predictive approach to thermodynamics and ultimately permits the estimation of Gibbs energy data. Entropy can be defined as the two equivalent definitions: The classical thermodynamic definition. For ionic hydrates, S degrees 298 /J K(-)(1) mol(-)(1) = 1579 (V(m)()/nm(3) formula unit(-)(1)) + 6 or = 2.621 + 6. A measure of an extent to which energy is dispersed is called entropy. The regression lines pass close to the origin, with the following formulas: For inorganic ionic salts, S degrees 298 /J K(-)(1) mol(-)(1) = 1360 (V(m)()/nm(3) formula unit(-)(1)) + 15 or = 2.258 + 15. The approach can also be extended to estimate entropies for hypothesized materials. V(m)() can be obtained from a number of possible sources, or alternatively density, rho, may be used as the source of data. 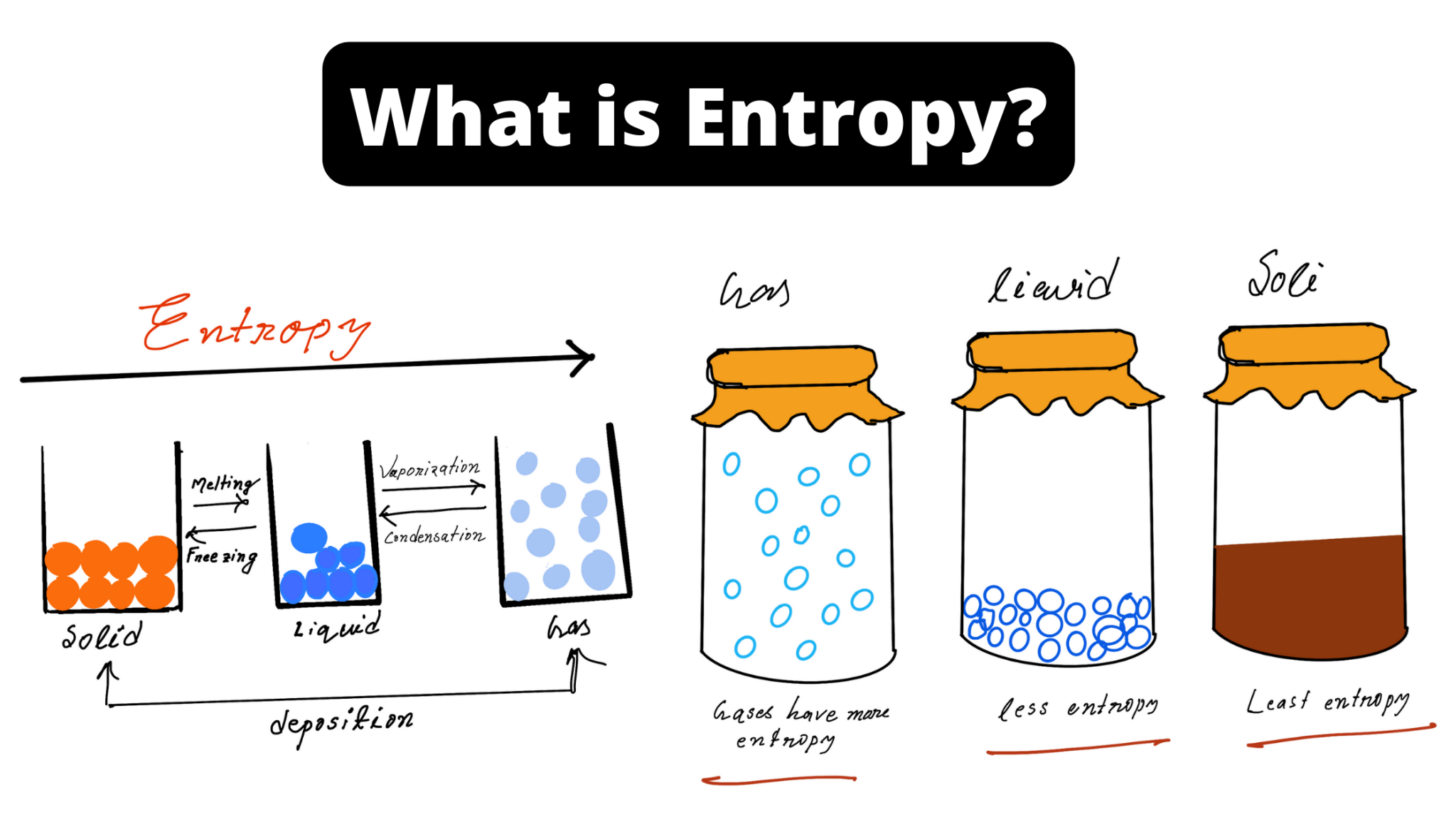
It is shown here that formula unit volume, V(m)(), can be employed for the general estimation of standard entropy, S degrees 298 values for inorganic materials of varying stoichiometry (including minerals), through a simple linear correlation between entropy and molar volume. 4 Absolute Entropy The absolute entropy of a substance may be calculated as the sum of the configurational entropy, the entropy changes associated with changes. The enthalpy and entropy change for the reaction are 30 KJ/mol and 105 J/K/mol, find out if T 285.7K. 
Standard absolute entropies of many inorganic materials are unknown this precludes a full understanding of their thermodynamic stabilities. The Enthalpy and entropy changes of a reaction are 40.63 KJmol1 and 108.8JK1mol1, the value of G is positive and hence the reaction is nonspontaneous.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
